Reviewed by Dr. Shradha Chakhaiyar, MS (OB-GYN), MRCOG (London), IVF & Laparoscopic Surgeon
Shradha IVF & Maternity, Patna, Bihar · 20+ Years Clinical Experience
📋 Table of Contents
- What Is Hysteroscopy?
- Why Standard Tests Miss Things
- HSG’s False-Negative Rate
- What Hysteroscopy Finds — 6 Conditions
- Diagnostic vs Operative Hysteroscopy
- When Is It Needed Before IVF?
- What the Research Shows
- What Happens During the Procedure
- Recovery — Day by Day
- How Soon Can I Start IVF After?
- C-Section, Asherman’s & Bihar Context
- Hysteroscopy at Shradha IVF, Patna
What Is Hysteroscopy?
Hysteroscopy is a minimally invasive procedure in which a thin, lighted camera called a hysteroscope — typically 2.9–5 mm in diameter — is inserted through the vagina and cervix into the uterine cavity, allowing direct real-time visualisation of the uterine lining. It is both diagnostic (examining the uterus for abnormalities) and operative (treating any abnormalities found, in the same sitting). No incisions are made. Most procedures take 10–30 minutes as day-care surgery. It is the gold standard for evaluating the inside of the uterus — superior to ultrasound and HSG for detecting intrauterine pathology.
The word hysteroscopy comes from the Greek: hystera (uterus) and skopein (to look). The principle is straightforward — look inside the uterus directly, with the full resolution of a camera rather than the indirect information provided by X-ray contrast or ultrasound waves. What makes it powerful for fertility medicine is its dual role: not only does it detect abnormalities that no other test reliably identifies, it can treat them immediately without the patient needing a second procedure.
In the context of IVF, hysteroscopy addresses the uterine side of implantation — a factor that is entirely invisible to embryo quality assessment, ovarian reserve testing, or semen analysis. A couple can have genetically perfect blastocysts ready to transfer and still fail to achieve pregnancy if the uterine environment is mechanically or biologically hostile. Hysteroscopy is the investigation that tells us whether the uterus is ready to receive those embryos.
The Problem with Standard Uterine Tests — Why HSG and Ultrasound Leave Gaps
To understand why hysteroscopy matters for IVF preparation, you first need to understand what the standard uterine tests actually measure — and what they consistently miss.
HSG’s False-Negative Rate — The Number That Changes the Conversation
HSG (hysterosalpingography) is the standard test for assessing tubal patency and providing a basic view of the uterine cavity. It involves injecting radio-opaque dye through the cervix and taking X-ray images as the dye flows through the uterus and out through the fallopian tubes. It is widely available, relatively inexpensive, and useful — but it has a significant limitation when it comes to detecting intrauterine pathology.
Multiple peer-reviewed studies have measured HSG’s diagnostic accuracy for intrauterine abnormalities using hysteroscopy as the gold standard:
- A study in Fertility and Sterility (2011) found HSG’s false-negative rate for intrauterine pathology was 78.43% — meaning that when HSG showed a normal uterine cavity, it was wrong about 78% of the time when significant pathology was subsequently found at hysteroscopy
- A PMC comparative study (2024) confirmed HSG had only 70% sensitivity for uterine cavity abnormalities overall, falling substantially lower for specific conditions like submucous fibroids and uterine septa
- A PMC meta-analysis specifically noted: “HSG is not able to diagnose polyps, septum, and submucous fibroids with significant accuracy. Therefore, hysteroscopy is indicated for confirmation.”
What does this mean for you in practical terms? If you had an HSG that showed a normal uterine cavity, there is a meaningful probability that a clinically significant abnormality — a polyp that could block your embryo from implanting, adhesions from a previous D&C or infection, or a small septum that increases miscarriage risk — was present but not detected. Your HSG being “normal” is reassuring evidence about your tubes. It is not reliable evidence that your uterine cavity is clear of pathology.
What Transvaginal Ultrasound Misses?
Transvaginal ultrasound (TVS) is excellent for assessing ovarian reserve (antral follicle count), detecting large fibroids and ovarian cysts, and measuring endometrial thickness. It is a blunt instrument for detecting small intrauterine pathology. Small endometrial polyps (under 1 cm), thin intrauterine adhesions, minor uterine septa, and chronic endometritis (inflammation of the lining) are systematically under-detected by standard TVS. Saline sonohysterography (SHG) — which involves instilling saline into the uterine cavity to separate the walls before ultrasound — performs better than plain TVS, achieving approximately 85–92% accuracy for intrauterine lesions, but is not universally available in India and still falls short of the direct visualisation that hysteroscopy provides.
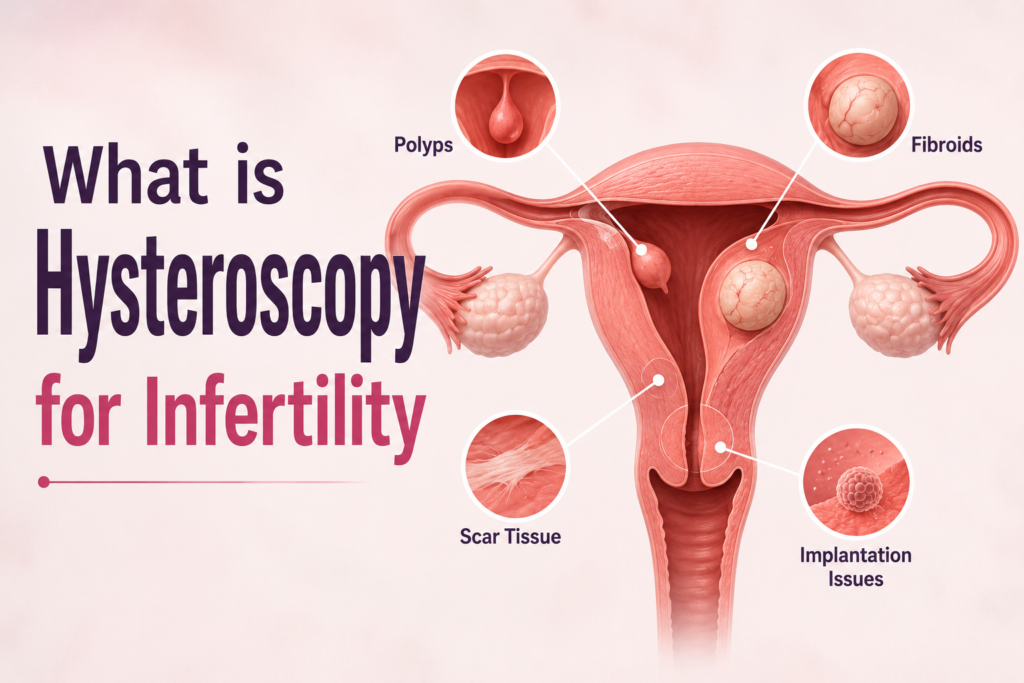
What Does Hysteroscopy Find? — The Six Conditions and Why Each One Matters for IVF
1. Endometrial Polyps
Endometrial polyps are soft, benign overgrowths of the uterine lining that project into the uterine cavity. They are the most common finding at hysteroscopy in infertile women — found in 15–30% of women with unexplained infertility. They impair implantation through several mechanisms: mechanical obstruction of the embryo’s attachment site, local inflammation that creates an immunologically hostile environment, and disruption of the endometrial receptivity signals (including LIF — leukaemia inhibitory factor) that guide implantation. Polyps below 1–2 cm are consistently missed on standard ultrasound and HSG. A 2024 study in the Journal of Clinical Medicine specifically confirmed that hysteroscopic polypectomy improves IVF outcomes in women with unexplained infertility. Removal is straightforward — the polyp is excised at the base using a resectoscope or grasping forceps in the same hysteroscopy session, leaving no scar and requiring no second procedure.
2. Submucosal Fibroids
Fibroids are benign smooth muscle tumours that grow within the uterine wall (intramural) or on the external surface (subserosal). The type that directly affects IVF outcomes is the submucosal fibroid — one that protrudes into the uterine cavity, distorting the endometrial surface. Submucosal fibroids that occupy more than 50% of the cavity are associated with a significantly reduced implantation rate — some studies report implantation rates of 4% versus 16% in controls. Hysteroscopic myomectomy (removal of the submucosal portion of the fibroid through the hysteroscope) is the standard treatment and significantly improves IVF outcomes when the fibroid is confirmed to be distorting the cavity. Critical point: intramural fibroids that do not distort the cavity are NOT improved by hysteroscopy — only the submucosal type justifies intervention, making precise preoperative classification essential.
3. Intrauterine Adhesions (Asherman’s Syndrome)
Intrauterine adhesions are bands of scar tissue that form between the walls of the uterine cavity, partially or completely obliterating the normal cavity. They are caused by any procedure that disrupts the endometrial lining: D&C (dilation and curettage), hysteroscopic surgery, C-section complications, severe uterine infections, or post-partum evacuation. Asherman’s Syndrome refers to more extensive adhesion formation. The adhesions prevent normal endometrial development, reduce the implantable surface area, and create a mechanical barrier to embryo attachment. They can be entirely asymptomatic — no change in periods, no pain — yet profoundly impair implantation. This is why they are so dangerous for IVF patients: a woman can feel completely normal yet have significant intrauterine scarring that will cause every embryo transfer to fail. Hysteroscopic adhesiolysis (surgical division of the adhesions) is the definitive treatment, usually requiring 1–2 procedures for mild-to-moderate disease and careful post-operative management for severe cases.
4. Uterine Septum
A uterine septum is a band of fibrous or muscular tissue that partially or completely divides the uterine cavity from the top downward — a congenital anomaly present from birth. Septae are associated with pregnancy loss (miscarriage rates of 26–94% depending on size), preterm labour, and implantation failure. The septum is poorly vascularised — embryos that implant on or near it are more likely to miscarry because the blood supply is insufficient for early placentation. Hysteroscopic metroplasty (surgical division of the septum) is a straightforward outpatient procedure that dramatically reduces miscarriage rates in women with this anomaly, typically restoring normal conception rates within 1–2 cycles post-surgery. Importantly, a septum that is clearly visible on 3D ultrasound may still require hysteroscopy for definitive confirmation and simultaneous treatment.
5. C-Section Niche Defect
A C-section niche (also called isthmocele) is a small pocket or defect at the site of a prior uterine incision from caesarean delivery. Menstrual blood and fluid can accumulate in this pocket, creating a local inflammatory environment that impairs sperm motility, embryo transport, and implantation in the adjacent endometrium. C-sections are estimated to occur in 20–60% of women who have a C-section delivery. In women with secondary infertility who are planning IVF, the niche may be a contributing cause of failure that is missed by standard investigations. This is particularly relevant in Bihar, where C-section rates in private hospitals are high, and many women with secondary infertility have a prior caesarean in their history. Hysteroscopy directly visualises the niche and allows assessment of its depth and clinical significance. Surgical correction options include hysteroscopic resection of the niche edges.
6. Chronic Endometritis
Chronic endometritis (CE) is persistent, low-grade inflammation of the endometrial lining — often without any symptoms, without abnormal discharge, and without any findings on ultrasound. It is caused by a low-level bacterial infection (most commonly from Enterococcus, Escherichia coli, or Streptococcus) that persists in the endometrium and creates an inflammatory environment hostile to implantation. CE is found in approximately 14–67% of women with recurrent implantation failure — a startlingly high prevalence that explains many unexplained IVF failures. Hysteroscopy reveals the characteristic appearance (stromal oedema, hyperaemia, and small endometrial polyps on CE), and an endometrial biopsy at the same time confirms the diagnosis. Treatment is a 2–3 week course of targeted antibiotics, after which pregnancy rates improve dramatically in affected women. CE is entirely invisible on ultrasound and HSG — it can only be reliably detected by direct uterine examination with biopsy.
What is the Difference Between Diagnostic and Operative Hysteroscopy?
| Type | What Happens | Duration | Anaesthesia | When Used |
|---|---|---|---|---|
| Diagnostic Hysteroscopy | The camera examines the uterine cavity only—no treatment performed. A biopsy may be taken. | 10–20 min | Local or none (office-based) | Evaluation before IVF, recurrent implantation failure workup, assessment after abnormal HSG |
| Operative Hysteroscopy | Camera examination plus treatment of any pathology found — polyp removal, adhesiolysis, septum resection, fibroid resection — in the same session. | 20–60 min | General or spinal (operating theatre) | When pathology is known or expected; when previous diagnostic hysteroscopy found a treatable abnormality |
| Office / Outpatient Hysteroscopy | Diagnostic ± minor operative steps using miniaturised instruments. No formal anaesthesia required. “See and treat” for small polyps. | 10–15 min | None or local analgesia | Increasingly preferred for first-line evaluation; small polyps can be removed in the same sitting |
When Is Hysteroscopy Treatment Needed Before IVF? — A Research-Based Decision Guide
This is the question at the heart of the topic — and it deserves an honest, nuanced answer rather than a simple “yes, always” or “no, not routinely.” The evidence, the guidelines, and the clinical reality are more complex than either extreme suggests.
What the Guidelines Say — ESHRE Position
It is important to present the full picture, including where the evidence is debated. The European Society of Human Reproduction and Embryology (ESHRE) — the world’s largest reproductive medicine organisation — states in its current guidelines that routine diagnostic hysteroscopy is not recommended as a standard investigation before the first IVF cycle in asymptomatic women with normal transvaginal ultrasound. Their reasoning: the individual RCTs on this question (including the inSIGHT trial published in The Lancet in 2016) did not show a statistically significant benefit when examined in isolation.
The tension between individual RCT data (which showed no significant benefit) and pooled meta-analysis data (which shows significant benefit) reflects the genuine uncertainty in this area. The JMIG 2023 meta-analysis — with its larger dataset and updated methodology — represents the most comprehensive analysis and does find benefit. But the scientific debate is real, and honest clinical guidance acknowledges it.
Dr. Shradha’s Clinical Framework — When We Recommend Hysteroscopy Before IVF at Shradha IVF
| Clinical Situation | Hysteroscopy Before IVF? | Rationale |
|---|---|---|
| Abnormal uterine cavity on HSG or ultrasound (polyp, fibroid, suspected adhesion) | Yes — essential before any IVF cycle | Known pathology must be treated before embryo transfer. No controversy here. |
| Unexplained infertility with normal HSG and normal TVS | Yes — strongly recommended before first IVF | HSG misses 40–78% of intrauterine pathology. Given the 2023 meta-analysis evidence, we believe the benefit-to-risk ratio strongly favours hysteroscopy. |
| One or more failed IVF cycles with good-quality embryos | Yes — essential before repeating IVF | 40% pathology detection rate in this group. Not doing hysteroscopy before a repeat cycle risks transferring into an environment that already failed for a treatable reason. |
| Secondary infertility, especially after C-section or D&C | Yes — Asherman’s and niche defect are real risks | Prior uterine procedures are the primary cause of intrauterine adhesions and niche defects. C-section history in Bihar = systematic hysteroscopy evaluation before IVF. |
| Recurrent miscarriage (2+ losses) | Yes — uterine septum and pathology evaluation | Uterine anomalies cause recurrent miscarriage. Hysteroscopy is part of every recurrent pregnancy loss workup. |
| Asymptomatic, normal TVS, normal HSG, first IVF attempt, no prior uterine procedures | Discussed with each couple individually | ESHRE does not recommend routine hysteroscopy here. The 2023 meta-analysis suggests benefit. We present the evidence and make the decision jointly with the couple, considering their age and how many IVF cycles they can tolerate. |
| Donor egg recipient, own uterus unstimulated | Yes — uterine cavity evaluation before recipient transfer | The recipient’s uterine environment is the variable. We evaluate it directly before the embryo transfer cycle. |
Step-by-Step Procedure: What Happens During Hysteroscopy Treatment?
Before the Procedure — Preparation
- The procedure is typically scheduled in the first half of the menstrual cycle (Day 7–14), when the endometrial lining is thinnest, and the uterine cavity is most visible
- Pre-operative blood tests (CBC, coagulation profile, blood group) and a recent ultrasound are usually required
- For office/diagnostic hysteroscopy: you may be asked to take ibuprofen or paracetamol 30–60 minutes before for pain management-no fasting required.
- For operative hysteroscopy under general anaesthesia: standard pre-operative fasting instructions apply (6–8 hours for food, 2 hours for clear fluids)
- A pregnancy test is performed before all hysteroscopy procedures
During the Procedure
- You lie in the same position as a gynaecological examination
- The hysteroscope is gently introduced through the vagina and cervix — no incisions are made
- The uterine cavity is slowly distended with saline or CO2 gas to open the space and allow visualisation
- The surgeon examines the entire uterine cavity on a monitor — the endometrial walls, fundus, tubal openings, and cervical canal — in real time
- If a polyp, adhesion, septum, or fibroid is found, it is treated immediately using miniaturised instruments (scissors, graspers, or resectoscope loop) passed through the hysteroscope’s working channel
- A biopsy of the endometrial lining may be taken to exclude chronic endometritis or endometrial hyperplasia
- The hysteroscope is gently withdrawn. No sutures are needed.
How to Recover After Hysteroscopy for Infertility Treatment?
Day 1 — Immediately After the Procedure
After diagnostic or minor operative hysteroscopy with local/no anaesthesia: mild-to-moderate cramping for 2–4 hours, similar to period pain. Light spotting or watery discharge. Most women can leave within 30–60 minutes and return to light activity the same day. After general anaesthesia: 2–4 hours of monitored recovery. Grogginess from anaesthesia. Arrange for someone to drive you home. Rest for the remainder of the day.
Day 2–3 — At Home
Period-like cramping continues but typically reduces significantly. Light spotting or discharge continues — this is normal endometrial tissue shedding. Paracetamol is adequate for most patients; ibuprofen is prescribed if necessary. Light activity is encouraged. Avoid tampons (pads only), swimming, sexual intercourse, and strenuous exercise.
Day 4–7 — Return to Normal Activity
Most patients return to desk work within 24–48 hours after diagnostic hysteroscopy, and within 3–5 days after minor operative procedures. Spotting typically resolves by Day 5–7. Sexual intercourse can usually resume after the spotting has stopped (approximately 1 week for diagnostic, 2 weeks for operative). Your surgeon will advise based on what was done.
Warning Signs — When to Contact Your Doctor Immediately
Contact Shradha IVF or go to the emergency if you experience: fever above 38.5°C, heavy vaginal bleeding (soaking more than one pad per hour for 2+ hours), severe or worsening abdominal pain, foul-smelling vaginal discharge, or difficulty passing urine. These are uncommon but warrant prompt assessment.
How Soon Can I Start IVF After Hysteroscopy? — Timing by What Was Found
This is the question every patient asks immediately after their procedure — and the honest answer depends on what was done, not a single universal timeline:
| Hysteroscopy Finding & Treatment | IVF Start Timing | Why This Interval |
|---|---|---|
| Diagnostic only — normal cavity, no treatment | Next menstrual cycle (3–5 weeks) | No healing required. IVF can begin from the next cycle. Some specialists start in the same cycle. |
| Small polyp removed (polypectomy) | 1–2 menstrual cycles (4–8 weeks) | The endometrium needs one cycle to re-epithelialise fully at the polypectomy site before it is ready for optimal implantation. |
| Submucosal fibroid removed (hysteroscopic myomectomy) | 2–3 menstrual cycles (8–12 weeks) | The fibroid base requires longer healing. A scan to confirm endometrial integrity before starting IVF stimulation is recommended. |
| Mild–moderate adhesiolysis (Asherman’s) | 2–3 cycles (8–12 weeks) with hormone treatment | Oestrogen supplementation post-adhesiolysis promotes endometrial re-growth over the divided adhesion sites. Repeat hysteroscopy may be needed to confirm cavity is clear. |
| Septum resection (metroplasty) | 1–2 cycles (4–8 weeks) | The resected septum heals relatively quickly. Confirm the normal cavity by scan before stimulation. |
| Chronic endometritis — diagnosed and treated with antibiotics | After completing antibiotic course + 1 cycle (4–6 weeks) | Complete the antibiotic course, allow one cycle for endometrial normalisation, then proceed to IVF. Repeat biopsy is sometimes done to confirm resolution. |
C-Section, Asherman’s Syndrome, and Failed IVF in Bihar — Dr. Shradha’s Clinical Perspective
I have seen this pattern dozens of times. The Asherman’s was the cause all along. It was simply never looked for. Bihar’s high C-section rate means this is not a rare story at our clinic — it is a pattern we address every week. If you have a prior C-section and a failed IVF, hysteroscopy is not optional at Shradha IVF. It is essential.
Bihar’s elevated C-section rate in private hospitals over the past two decades has created a generation of women — now in their 30s, trying for second pregnancies or undergoing IVF for the first time — with potential intrauterine scarring from their deliveries. This is not hypothetical. C-section creates a scar in the lower uterine segment. In some cases, this extends into the cavity. In others, post-operative inflammation and healing create adhesions. Post-partum infections — which may receive inadequate antibiotic treatment in rural Bihar — compound this risk significantly.
The consequences are invisible on standard tests but clinically devastating for IVF outcomes. An Asherman’s that reduces the functional endometrial surface by 30% may produce a normal-appearing ultrasound lining thickness. The HSG may show contrast reaching both tubes. And then every embryo transfer fails — not because of the embryos, not because of the protocol, but because the cavity it was transferred into was physiologically hostile to implantation in a way that no standard pre-IVF test detected. Hysteroscopy is the test that finds this. It is why we do it.
Hysteroscopy Treatment at Shradha IVF & Maternity in Patna — What to Expect
At Shradha IVF, hysteroscopy treatment is performed by Dr. Shradha Chakhaiyar — an MRCOG-qualified specialist with over 20 years of gynaecological surgery experience in Bihar. Both diagnostic and operative hysteroscopy are available as day-care procedures in our fully equipped operating suite.
- Combined Hystero-Laparo: When both uterine cavity (hysteroscopy) and pelvic assessment (laparoscopy) are needed, both procedures can be performed simultaneously under a single general anaesthetic — reducing the total number of procedures, recovery periods, and surgical exposures for the patient. This combined approach is our standard recommendation for unexplained infertility cases where both internal and external pelvic pathology needs evaluation.
- Hysteroscopy cost at Shradha IVF, Patna: Diagnostic hysteroscopy typically ranges from approximately ₹15,000–₹35,000. Operative hysteroscopy (including polyp removal, adhesiolysis, or septum resection) typically ranges from ₹35,000–₹75,000 depending on complexity. A complete cost estimate is provided before any procedure begins.
- Results available immediately: Unlike HSG or biopsy results that take days, hysteroscopic findings are visible in real time on the monitor. We discuss what was found with the patient at the same post-procedure appointment and plan the next steps — whether that is immediate IVF preparation or a recovery period before stimulation — before the patient leaves the clinic.
- EMI options: Hysteroscopy is included in the treatment procedures available on Shradha IVF’s EMI payment plan for couples who need financial flexibility.
FAQs Related to Hysteroscopy for Infertility
Yes, hysteroscopy can help improve fertility in some cases by identifying and treating problems inside the uterus that may affect conception. Conditions such as uterine polyps, fibroids, scar tissue, adhesions, or uterine abnormalities can interfere with implantation or pregnancy. Correcting these issues may improve the chances of natural conception or IVF success.
A hysteroscopy is performed to examine the inside of the uterus and diagnose or treat uterine conditions. Doctors commonly use it to investigate abnormal bleeding, infertility, repeated miscarriages, uterine polyps, fibroids, adhesions, or structural abnormalities.
It depends on the type of hysteroscopy being performed. Diagnostic hysteroscopy is often done while the patient is awake and may involve minimal discomfort. Operative hysteroscopy, where treatment or surgical procedures are performed, may require local, regional, or general anesthesia.
A hysteroscopy with dilation and curettage (D&C) usually takes around 15–30 minutes, although the total hospital visit may take longer due to preparation and recovery time.
A hysteroscopy is not required for everyone. Doctors may recommend it when symptoms such as abnormal uterine bleeding, infertility, recurrent pregnancy loss, or suspected uterine abnormalities need further evaluation or treatment.
The recommended waiting period depends on the procedure performed. Many women can start trying to conceive after one menstrual cycle, while others may need to wait longer if extensive treatment was done. Your doctor will provide guidance based on your specific condition.
Yes, many women can conceive naturally after hysteroscopy, particularly if the procedure corrected issues affecting fertility such as uterine polyps, adhesions, or fibroids. Pregnancy chances vary depending on age, reproductive health, and any underlying fertility factors.
Your Uterus Deserves to Be Seen — Not Just Screened.
At Shradha IVF & Maternity in Patna, Dr. Shradha Chakhaiyar evaluates the uterine cavity directly — giving your IVF cycle the best possible start by ensuring the environment your embryo will be transferred into has been confirmed ready, not just assumed normal.
Book Your Consultation at Shradha IVF →